How it works
questhealth.com offers 150+ consumer-initiated Quest Diagnostics lab tests to empower you to have more control over your health journey. Choose from a variety of test types that best suit your needs.
 HSA/FSA Accepted
HSA/FSA Accepted
questhealth.com offers 150+ consumer-initiated Quest Diagnostics lab tests to empower you to have more control over your health journey. Choose from a variety of test types that best suit your needs.
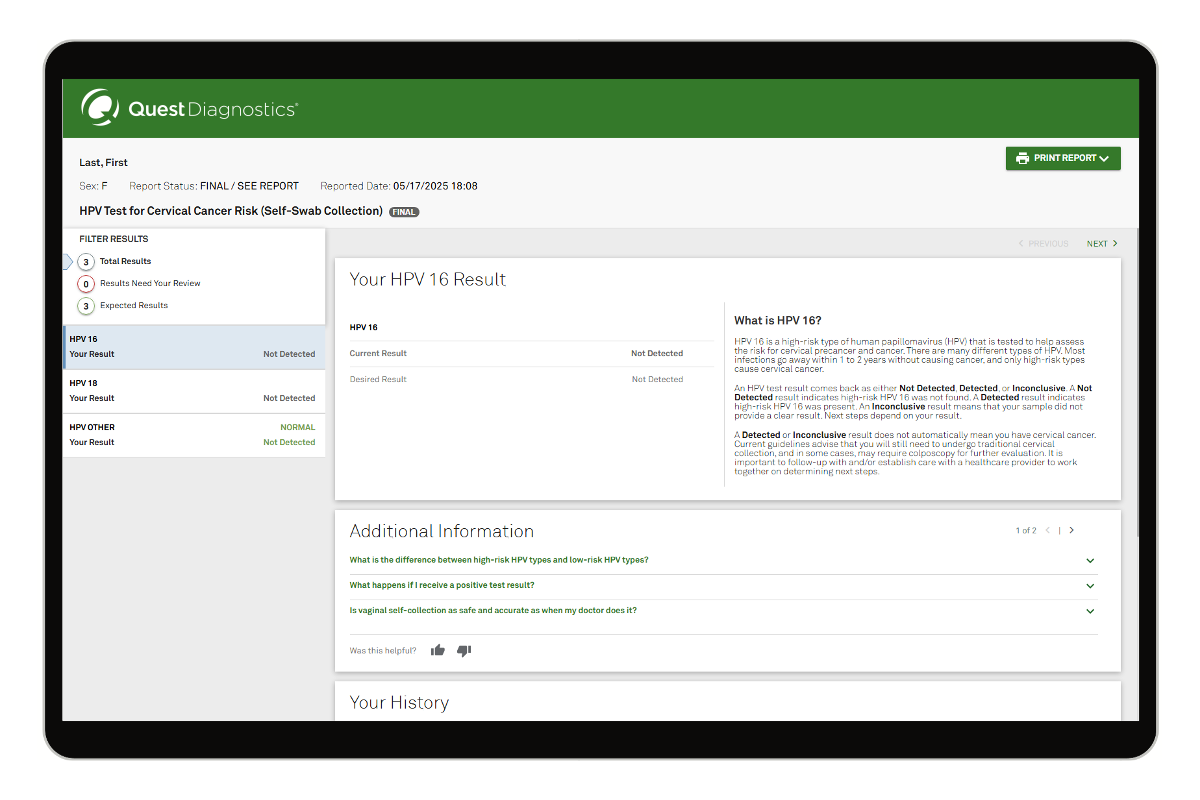
Yes. According to the CDC, by following the step-by-step collection instructions, patients can properly collect their own samples, with test results that are as accurate when they are collected by a doctor.4
If you have questions prior to performing your self-collection, you may ask the Patient Service Representative (PSR) or call 1-833-700-2423. The help line is open Monday-Friday 8:00 AM to 7:00 PM EST. It is closed on weekends and holidays. If you are unable to self-collect, please contact your doctor. The collection cannot be done by the PSR.
You may be able to use your FSA or HSA funds to purchase lab tests at questhealth.com. Please review your FSA/HSA plan or check with a tax professional to confirm specific requirements for individual eligibility.
Tests purchased on questhealth.com are intended for individuals paying directly—we do not bill insurance, and purchases cannot be submitted for insurance reimbursement.
If your doctor ordered a lab test for you and you are looking to book an appointment for sample collection, you can schedule an appointment here https://appointment.questdiagnostics.com/as-home.
For other questions related to a lab test your doctor ordered for you, including billing and results, you can visit the main Quest Diagnostics site here https://www.questdiagnostics.com.
ACOG. Cervical Cancer Screening. Accessed May 13, 2025. https://www.acog.org/-/media/project/acog/acogorg/womens-health/files/infographics/cervical-cancer-screening.pdf?rev=2ca474997ed747a5801de351a35c2c3e&hash=B037FB71487B9F31ECB77C833E7A75F7
US Preventive Services Task Force. Final recommendation statement. Cervical cancer: screening. August 21, 2018. Accessed December 20, 2024. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/cervical-cancer-screening
American Cancer Society. The American Cancer Society guidelines for the prevention and early detection of cervical cancer. 2024. Updated April 22, 2021. Accessed May 13, 2025. https://www.cancer.org/cancer/types/cervical-cancer/detection-diagnosis-staging/cervical-cancer-screening-guidelines.html
CDC Preventing Chronic Disease. Self-Collection for Primary HPV Testing: Perspectives on Implementation From Federally Qualified Health Centers. Accessed March 24, 2026. https://www.cdc.gov/pcd/issues/2023/23_0056.htm
Inturrisi F, Aitken CA, Melchers WJG, et al. Clinical performance of high-risk HPV testing on self-samples versus clinician samples in routine primary HPV screening in the Netherlands: an observational study. Lancet Reg Health Eur. 2021;11:100235. doi:10.1016/j.lanepe.2021.100235
Yoda R, Cherne R, Lin J, et al. Validating human papillomavirus self-collection testing as an alternative approach to cervical cancer screening. J Am Soc Cytopathol. 2024;13(5):S25-S26. Accessed February 28, 2025. https://doi.org/10.1016/j.jasc.2024.08.044
Le A, Rohweder C, Wheeler SB, et al., Self-collection for primary HPV testing: perspectives on implementation from federally qualified health centers. Prev Chronic Dis. 2023;20:E93. doi: 10.5888/pcd20.230056
Danan ER, Amundson EC, Gowdy-Jaehnig A, et al. “It feels like health care with the patient in mind”: VA patient and staff perspectives on self-collected HPV testing. Women’s Health Issues. 2024;34(5):518-527. doi:10.1016/j.whi.2024.05.003
Goldstein A, Plafker B, Stamper S, et al. Patient satisfaction with human papillomavirus self-sampling in a cohort of ethnically diverse and rural women in Yunnan Province, China. J Low Genit Tract Dis. 2020;24(4):349-352. doi:10.1097/LGT.0000000000000560
Fontenot HB, Fuzzell L, Brownstein NC, et al. Health care provider willingness to recommend self-collected tests for human papillomavirus: a mixed methods examination of associated factors. Womens Health Issues. 2024;34(5):506-517. doi:10.1016/j.whi.2024.05.005
Quest Diagnostics. HPV DNA (16, 18, Other High Risk), PCR, Vaginal Self-Collected. Accessed March 24, 2026. https://testdirectory.questdiagnostics.com/test/test-detail/14263/hpv-dna-16-18-other-high-risk-pcr-self-collected?p=r&q=14263&cc=PHP
National Cancer Institute Cancer Trends Progress Report. Cervical Cancer Screening. Accessed August 9, 2024. https://progressreport.cancer.gov/detection/cervical_cancer
Subramaniam A, Fauci JM, Schneider KE, et al. Invasive cervical cancer and screening: what are the rates of unscreened and underscreened women in the modern era? J Low Genit Tract Dis. 2011;15(2)110-113. doi: 10.1097/LGT.0b013e3181f515a2
MedpageToday. U.S. Sees ‘Troubling’ Trend in Cervical Cancer Screening Rates. Accessed March 7, 2025. https://www.medpagetoday.com/obgyn/cervicalcancer/114506
CDC Sexually Transmitted Infections (STIs). About Genital HPV Infection. Accessed March 24, 2026. https://www.cdc.gov/sti/about/about-genital-hpv-infection.html#cdc_disease_basics_symptoms-signs-and-symptoms
National Cancer Institute. HPV and Cancer. Accessed March 24, 2026. https://www.cancer.gov/about-cancer/causes-prevention/risk/infectious-agents/hpv-and-cancer
National Cancer Institute. Cervical Cancer Prognosis and Survival Rates. Accessed March 24, 2026. https://www.cancer.gov/types/cervical/survival
U.S. Department of Health and Human Services Healthy People 2030. Increase the proportion of females who get screened for cervical cancer — C-09. Accessed March 24, 2026. https://odphp.health.gov/healthypeople/objectives-and-data/browse-objectives/cancer/increase-proportion-females-who-get-screened-cervical-cancer-c-09
American Cancer Society. Types of HPV. Accessed March 24, 2026. https://www.cancer.org/cancer/risk-prevention/hpv/types-of-hpv.html
CDC Human Papillomavirus (HPV). About HPV. Accessed March 24, 2026. https://www.cdc.gov/hpv/about/index.html